[Editor’s note: This transcript was generated using both transcription software and a human’s light touch. It has been edited for style and clarity.]
Mary Agnes Carey: Hello from ั๎น๓ๅ๚ดซรฝา๎l Health News and WAMU radio in Washington, D.C. Welcome to What the Health? I’m Mary Agnes Carey, managing editor of ั๎น๓ๅ๚ดซรฝา๎l Health News, filling in for Julie Rovner this week. And as always, I’m joined by some of the best and smartest health reporters covering Washington. We’re taping this week on Friday, April 17, at 10 a.m. As always, news happens fast and things might have changed by the time you hear this. So here we go.
Today we’re joined via videoconference by Anna Edney of Bloomberg News.
Anna Edney: Hi, everybody.
Carey: Joanne Kenen of the Johns Hopkins Bloomberg School of Public Health and Politico Magazine.
Joanne Kenen: Hi, everybody.
Carey: And my ั๎น๓ๅ๚ดซรฝา๎l Health News colleague Emmarie Huetteman.
Emmarie Huetteman: Hey there.
Carey: Later in this episode, we’ll play Julie’s interview with immigration attorney Michelle Canero about the impact the Trump administration’s immigration policies are having on the medical workforce. But first, this week’s news โ and there is plenty of it.
On Thursday, President [Donald] Trump nominated Dr. Erica Schwartz to lead the Centers for Disease Control and Prevention. Schwartz, a vaccine supporter, served as a deputy surgeon general in President Trump’s first term, and during the coronavirus pandemic she ran the federal government’s drive-through testing program. She’s also a Navy officer and a retired rear admiral in the Commissioned Corps of the U.S. Public Health Service. Her appointment requires Senate confirmation. President Trump also announced other changes to the agency’s top leadership: Sean Slovenski, a health care industry executive, as the agency’s deputy director and chief operating officer; Dr. Jennifer Shuford, health commissioner for Texas, as deputy director and chief medical officer, and Dr. Sara Brenner, who briefly served as acting commissioner of the FDA [Food and Drug Administration], as a senior counselor to Department of Health and Human Services Secretary Robert F Kennedy Jr. So we’ve discussed previously on the podcast several times that the CDC has lacked a permanent director for most of the president’s second term. Will Dr. Schwartz, if confirmed, and the other members of this new leadership team make the difference?
Huetteman: I think that we’ve seen a CDC that’s been in a protracted period of turmoil, and this is going to be an opportunity for maybe a shift in that. Dr. Schwartz would actually be the agency’s fourth leader in a little more than a year, and we’ve talked on the podcast about how naming someone who could fit the bill to lead the CDC was a difficult task facing the Trump administration. They needed someone who could support the MAHA [Make America Healthy Again] agenda while not embracing some of the more anti-vaccine views, and that person needed to be able to win Senate confirmation, which isn’t a given, even with this Republican-controlled Senate.
Edney: And I think we’ve seen that there have been some people already in the MAHA coalition that have come out and been upset about this pick. So I think what that shows is a calculated decision by the administration to, kind of, as they’ve been doing for this year, is kind of not focus on the vaccine part of Secretary Kennedy’s agenda and to, as Emmarie said, try to get someone that can get through Senate confirmation. We’ve already seen the surgeon general nominee be held up in the Senate because she was not as strong on vaccines as I think some would have liked to see when she had her confirmation hearing.
Kenen: So this happened late yesterday, and I’ve been traveling this week, but I did have a chance to talk to some public health people about her, and there was sort of this audible sigh of relief. The Senate is a very unpredictable place, and we live in very unpredictable times. At this point, my initial gut reaction is she’s got a pretty good chance of confirmation. The other thing, I think some of the other appointees, there’s a little bit more concern about, but what really matters is who is the face of the CDC, and she would be the face of the CDC. She would be in charge, and people like her. Also, this is an administration that has not had a lot of minorities, and she will be, she’s a Black woman. respected in her field. And that also is going to โ she needs to be able to speak to all Americans about their health, and I think that people welcome that as well, both her credentials and her life experience. So, yeah, I think that MAHA is sort of in this funny moment now, because clearly Kennedy isn’t doing everything that people wanted or expected. And so we’ll sort of see how the โ I think if he had his ideal CDC director, this, we can probably surmise that this would not, she would not be the first on his list. But there’s a certain amount of adaptation going on at the moment. So I think many, many people will be relieved to see somebody get through, confirmed pretty quickly. People can get held up for things that have absolutely nothing to do with the CDC or public health. The Senate has all sorts of peculiarities. But I think there’s probably going to be a desire to get this done pretty quickly.
Carey: All right. Well, we’ll see what happens, and we will go back to the MAHA folks a little bit later in the podcast. But right now I want to shift to Capitol Hill. Thursday was a very big day on the Hill for HHS Secretary Kennedy. He kicked off a series of appearances before Congress. This week he’s testifying before three House committees before he heads over to the Senate next week. This is the first time that the secretary has visited some of these House panels, and while the purpose of the latest congressional visit is to talk about President Trump’s HHS budget request, this also was the first time that a lot of lawmakers ever had an opportunity to talk to Kennedy, and what they asked him sometimes deviated, maybe quite a bit, from that subject of federal funding. The topics included Medicaid fraud, measles outbreaks, the birth-dose recommendation for the hepatitis B vaccine, peptides, unaccompanied minors, and more โ actually, much more when you look at the hearings from yesterday, and I’m sure that will also happen with today’s session. What stood out to you about Kennedy’s testimony this week?
Edney: I think it was the mix of questions, and you sort of alluded to this, but they wanted, the members of Congress wanted to talk about so many things. And I feel like in the earlier hearing, which was in the House Ways and Means Committee, that it was, there was a lot of focus in the beginning on fraud, and that sort of surprised me, and then we saw maybe one or two questions on vaccines. And so I thought the mix of questions, the things that members were interested in, were really interesting. And it did โ there were some fiery moments, but for his first time on the Hill in a while, for such a controversial Cabinet member, I thought they were pretty tame.
Kenen: Yeah, I watched a fair amount of the morning. I did not see the afternoon, but I read about the afternoon, and I totally agree with Anna’s take. This administration and Kennedy did what this administration has been doing. They blame all problems on [former president Joe] Biden and the prior administration. And to be fair, Democrats, when they’re in power, they, I don’t think they do it quite to this extreme, but Democrats spend, when they have the chance, they blame things on Republicans. So that’s sort of Washington as usual. The emphasis on fraud has been a hallmark of this administration, particularly in health and social services. And you’ve seen, of course, in the way they’ve gone after blue states in particular. And a lot of their justification for the changes in Medicaid that are coming in the coming year are supposedly because of massive fraud and they’re cracking down. It was not dominated by vaccines, and I was watching Kennedy’s face really carefully. When he was asked about the first child to die of measles in Texas last year, and a Democrat asked him could the vaccine have saved her life, and you could sort of see him just, you just sort of watch his facial expressions, and he knew he had to say this, and he came out with the word “possibly,” and, which is a change. And then in the afternoon โ where I did not, as I said, I did not watch the afternoon, but I read about it โ he was much more certain. He was much stronger about the measles vaccine and said it’s, the measles vaccine, is safer than measles, which is a big signal shift there.
Huetteman: It’s true, although I will point out, though, that he did stand by the decision to remove the recommendation for the birth dose of the hepatitis B vaccine when he was pressed on that. So it was, I agree it was a softening, I’d say. At least it wasn’t a dramatic turnaround from what he’d said or not said in the past. But for him, it was at least a softening.
Kenen: In the hepatitis B recommendation, he said that the biggest threat to infection was at, through birth, at, through the mother, and if you test the mother, the baby is not at risk. And that’s partially true, and that is a significant factor to eliminate risk. It doesn’t โ it minimizes risk. It does not eliminate risk. Babies can and have been infected in the first weeks of life in other ways. The recommendation was not to totally eliminate that vaccine. It was to postpone it. But there’s, public health, still believe that, in general, many public health leaders would still say that the vaccine at birth is the better way of doing it.
Carey: The focus was, theoretically, on the budget request from the administration. Did the secretary shed any light on those priorities or their impacts? I was taken, I think in the afternoon hearing I read about various lawmakers, including Rosa DeLauro from Connecticut, who sort of just said: A CDC cut of 30%? We’re not gonna do that. And there were also some Republican members who jumped in to sort of say, I don’t think we’re going to do the cuts you envision. But did the secretary defend them? Did he bring any new clarity to them?
Edney: I don’t feel like I gained any new clarity on it. I think to bring it back to Budget 101, I guess, is like when the president, when the administration, sends down their budget, I think a lot of people already assume it’s dead on arrival. And maybe even though Kennedy is there to talk about the budget, it does become this broader hearing, because they don’t get him on the Hill that often and people go there to talk about all kinds of things, and I think that he probably knew that he didn’t have to defend it in the same way, because it’s not going to happen.
Carey: Sure. As they say, the president proposes and Congress disposes. But Joanne, you want to jump in?
Kenen: Yeah, there’s something significant about this administration, which is Congress has repeatedly authorized more money for various health programs and science programs, and the administration doesn’t spend it, so that there’s a different dynamic. Traditionally, yes, Congress โ the president proposes, Congress legislates, and then people go off and spend money. That’s what people like to do. And in this case, when Congress has, in a bipartisan way, differed with the administration and restored funding, it hasn’t all gone, those dollars haven’t gone out the door. So the entire sort of checks-and-balances system has been askew in terms of funding. I agree with everybody here. I do not think that Congress is going to accept these extreme cuts across the board in health care and health policy, in public health and science and NIH [the National Institutes of Health] and everything, but I don’t know what they’re actually going to spend at the end of the day.
Carey: Emmarie, you wanted to jump in.
Huetteman: Yeah, there was one striking exchange to me where the secretary acknowledged he wasn’t happy with the cuts that were proposed. I think those were his words. But he pretty quickly added, and neither is President Trump, and he framed it as a matter of making hard decisions when faced with federal budget shortfalls.
Carey: All right. Well, we’ll keep watching this as it moves through Congress. Also during yesterday’s House Ways and Means hearing, some Democrats took issue with past statements from Secretary Kennedy and President Trump that linked Tylenol use during pregnancy to autism in children. released this week in JAMA Pediatrics found that the use of Tylenol by women during pregnancy was not associated with autism in their children. This nationwide study from Denmark followed more than one and a half million kids born between 1997 and 2002, including more than 31,000 who were exposed to Tylenol in the womb. in another medical journal examining community water fluoridation exposure from childhood to age 80 found no impact on IQ or brain function. Kennedy has claimed that fluoride in water has led to IQ loss in children. These studies clearly debunk medical claims that have gotten a lot of attention. Will these findings have an impact now?
Kenen: I think we’ve seen over and over and over again that there are people who are very deeply wedded to certain beliefs, and new science, new research, does not deter them from those beliefs. We also see some people who are sort of in the middle, who are uncertain, and new findings can shift their beliefs, right? And then, of course, there’s a lot of โ these are not new studies. I mean these are new studies but they are not the first of their kind. The reason we’ve been using fluoride for, what, 60 years now in the water. Tylenol has been around a long time. So is it going to change everybody’s belief? No. Is it going to perhaps slow the push to ban fluoridation? Perhaps. But I just don’t think we know, because we’re sort of on these dual-reality tracks regarding a lot of science in this country, where once people sort of buy into disinformation, they’re very, it’s very hard to change โ or misinformation โ it’s hard to change people’s minds.
Edney: I do think, on the Tylenol front โ I absolutely agree with what Joanne said overall. And I think on the Tylenol front that it’s possible that this study will give pediatricians something to give and talk about with parents that are asking. I think there still is some confusion among some people. It’s not a huge, I don’t think, widespread thing, but I think there are some new parents who are wondering. And if you are able to take this study that is published in 2026 โ it just happened, it was after Trump made his statements โ I think maybe that would give them something to talk about with their patients.
Kenen: I agree with Anna. I think the Tylenol one is easier to change than some of the fluoridation stuff going on, partly because so many of us โ and we should just say, it’s not just the Tylenol, the brand. It’s acetaminophen, which I’ve never pronounced right. I think those of us who have been pregnant, we’ve taken that in our life before and we don’t think of it as a big, dangerous, heavy prescription drug. I think we’ve, it’s something we feel comfortable with. And I think there’s also the counterinformation, which is, a fever in a pregnant woman can, a pregnant person can be dangerous to the fetus. So I think that one’s a little โ and I don’t, also, I don’t think it’s as deep-rooted. The fluoridation stuff goes back decades, and the Tylenol thing is sort of new. And it might be, I’m not sure that the course of these arguments โ I think that Tylenol is easier to counter than some other things, because partly just we do feel safe with it.
Carey: All right. We’re going to take a quick break. We’ll be right back.
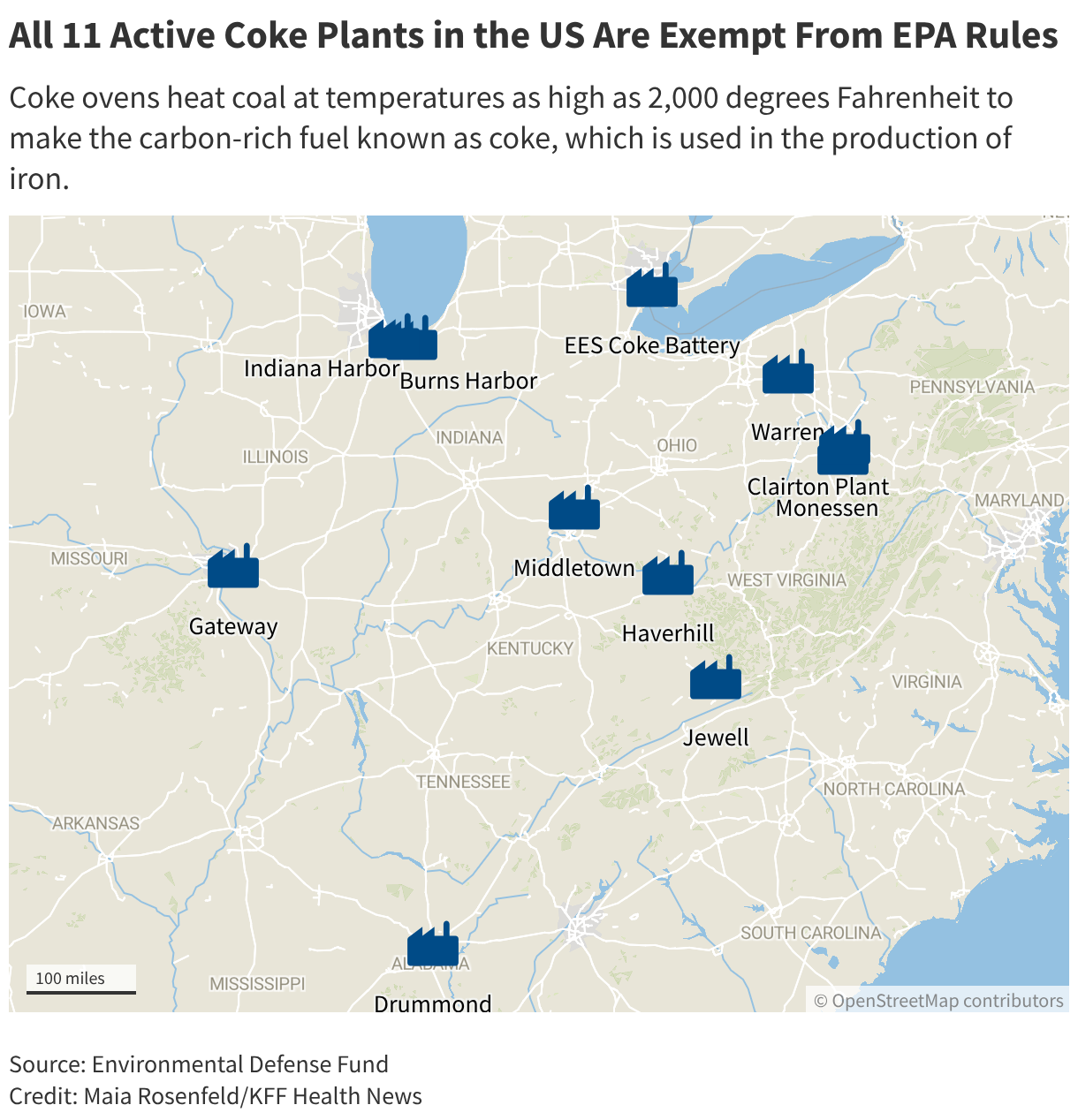
We’re back and talking about how the Trump administration is managing the voters behind the Make America Healthy Again, or MAHA, movement, which helped President Trump win the 2024 election. My colleagues Stephanie Armour and Maia Rosenfeld wrote about the administration’s recent decision to give coke oven plants in the U.S. a one-year exemption from tougher environmental standards. And that was a move that angered some MAHA activists who wondered if the GOP is more beholden to industry than the MAHA agenda. President Trump, HHS Secretary Kennedy, and other top administration officials met recently at the White House with a group of MAHA leaders to calm concerns that the administration is moving too slowly on food policy changes, and they are concerned about the president’s recent support of the pesticide glyphosate. According to press reports, the MAHA folks seem to feel their concerns were heard during that session. But is this ongoing conflict between the president and this key political constituency, will it be one that keeps brewing as the midterm elections approach?
Edney: Yes, 100%. I think it will continue to brew. I think that meeting was thrown together so quickly that some members of the MAHA movement who were invited couldn’t even make it. So it wasn’t exactly a long-planned, seemingly deep desire to fix everything. But it was, as you’ve said, an effort to kind of hear them out and make them feel heard. No one that I’ve talked to has said everything is fixed now. It’s more of a to-be-determined We will see what the administration will do moving forward, if they will listen to any of our plans โ which we will not share with you, by the way โ to make us happy. And I think that that’s going to continue. There’s a rally planned in front of the Supreme Court on glyphosate later this month where a lot of those people will be, and so I think that they’re upset and they’re stirring up, that concern is only going to get stirred up more.
Carey: Emmarie.
Huetteman: It’s a small thing, but our fellow podcast panelist Sheryl Stolberg at The New York Times during this White House meeting where President Trump was meeting with MAHA leaders, one of the leaders made a joke about how this is not a group that’s going to be, quote, “Team Diet Coke,” and the president apparently took that as a cue to press that Diet Coke button he famously has on his desk and summon a server who apparently brought him a Diet Coke. Supporters of MAHA have been clear that they want not just for the Trump administration to promote policies supporting priorities like healthy eating and removing food dyes, but also they want them to rein in or end policies they don’t support. And that weed-killer executive order, that really was a big example of that. The MAHA constituency made it clear that they felt betrayed by that order, and they’re going to have to do some work to walk that back.
Carey: We’ll also see how, with their concerns about the new CDC director nominee, which they’re already voicing, we’ll see how that plays out.
Kenen: No, I just think that we are, as we mentioned at the beginning, we’re seeing cracks, right? We’re seeing โ none of us are privy to any conversations that President Trump has had privately with Secretary Kennedy. But his, Secretary Kennedy’s, public statements have been a little different than they were a few months ago. There’s certainly been reports that he’s been told to soft-pedal vaccines and talk about some of the things that there’s more unanimity across ideological and party lines. Healthier food โ there’s debate about how to, whether, there’s debate about how Kennedy defines healthier food. But in general, should we eat healthier? Yes, we should eat healthier. Should our kids get more exercise? Yes, our kids should get more exercise. Do we have too much chronic disease? Yes, we have too much chronic disease. So they’re sort of this, trying to move a little bit more, sort of this sort of top line, very hazier agreement. But at the same time, the people who are sort of really the core of MAHA, as Kennedy has sort of created it or led it, there’s cracks there.
Carey: All right, we’ll see. We’ll see where that goes. But let’s go ahead and move on to ACA enrollment. A found that 1 in 7 people who signed up for an Affordable Care Act plan failed to pay their first month’s premium. The analysis from Wakely consulting group found that nationally around 14% of those who enrolled in ACA plans didn’t pay their first bill for January coverage. Now we know the elimination of the enhanced ACA tax credits and higher premium costs led to lower enrollment in the ACA exchanges, with sign-ups for 2026 falling to 23 million from 24 million a year ago. But how do you interpret this finding that 14% of enrollees didn’t pay their January premium? Is it a sign of more trouble ahead?
Edney: I think it could be a sign of more trouble ahead. Some โ what we’re seeing is sticker shock. And there may be some people who are trying to deal with that and won’t be able to as the months go on. And so, yeah, I think it could mean that even more drop out, and that means more people lose coverage and are uninsured.
Kenen: I think there was sort of a general, initial, misleading sigh of relief when in December, when the enrollment figures, the drop wasn’t as bad as some feared. But at the same time, people said: Wait a minute. This doesn’t really count. Signing up isn’t the same thing as staying covered. The drop in January was significant, we now know. And I agree with Anna. I think we don’t know how many more people will decide they can’t afford it. Or we don’t know whether the big drop is January. Probably a lot of it is, because you get that first bill. But can, will more people drop? Probably. We have no way of knowing how many. And it also depends on the economy, right? If more people lose jobs, right now it’s still pretty, kind of still pretty stable, but we don’t know what’s ahead. We don’t know what’s going to happen with the war. We don’t know many, many, many โ we don’t know anything. So the future is mysterious. I would expect it to drop more. I don’t think, I don’t know whether this is the big drop or February will be just as bad. I suspect January will be the biggest. But who knows? It depends on other outside factors.
Huetteman: We’re also seeing a drop-off in the kind of coverage that people are choosing. That analysis that you referenced, Mac, showed that there was a 17% drop in silver plan membership, with most of those folks switching to bronze plans, which, in other words, that means they switch to plans that have lower monthly premiums but they have higher deductibles. And that means that when you get sick, you owe more, in some cases much more, before your insurance starts picking up the tab. And I think really what this means is people are more exposed to the high charges for medical services, bigger bills when you get sick. I think that
Kenen: I think that the Republicans were seen as having pushed back a lot of the health impacts of the so-called One Big Beautiful Bill and that it would be after the election. And I and others wrote: No, no, no, no, no. We’re going to see this playing out before the election. This is a really big political red flag, right? This is a lot more people becoming uninsured, which makes other people worried about their insurance and stability. So I think this is definitely going to โ it may not be. There are other things going on in the world. Health care may not be the dominant theme in this year’s election. But yes, this is going to be, the off-year elections are going to be health care elections, like almost every one else has been forโ
Carey: Oh yeah.
Kenen: โsince the Garden of Eden, right?
Carey: Absolutely, it’s a perennial. All right, we’ll keep our eye on that. That’s this week’s news. Now we’re going to play Julie’s interview with immigration attorney Michelle can arrow, and then we’ll be back with our extra credits.
Julie Rovner: I am pleased to welcome to the podcast Michelle Canero. Michelle is an immigration attorney from Miami and a member of the board of Immigrants’ List, a bipartisan political action committee focused on immigration reform. Michelle, thanks for joining us.
Michelle Canero: Thank you for having me.
Rovner: So, we’ve talked a lot about immigration policy on this podcast over the past year, but I want to look at the big picture. How important to the U.S. health care system are people who originally come from other countries?
Canero: I think the statistics speak for themselves. One in three residency positions can’t be filled by American graduates alone. That means 33% of these residency positions are being filled by immigrant workers. Twenty-seven percent of physicians are foreign-born. Twenty percent of hospital workers are immigrants. And, at least in Florida, a large percentage of our home health care workers happen to be immigrants. And we depend on this population heavily in the health care sector.
Rovner: Now, we talk a lot about the Trump administration’s crackdown on illegal immigration, but we talk a little bit less about their sort of messing with the legal immigration system. And there’s a lot going on there, isn’t there?
Canero: There is. And I think that the campaign talking points were illegal immigration but what we’re actually seeing is a little more sinister. I think that the goal of leadership at the head of DHS [the Department of Homeland Security] and DOS [the State Department], or really Stephen Miller, is pushing something called reverse migration, which is really not about limiting illegal immigration but reducing the immigrant population in the United States. And I think that’s where the real concern is and why you’re seeing these policies that directly affect legal immigrants.
Rovner: We talk a lot about doctors and nurses and skilled, the top skilled, medical professionals who make up a large chunk of the United States health care workforce. We don’t talk as much about the sort of midlevel professional workers and the support staff. They’re also overwhelmingly immigrant, aren’t they?
Canero: Yeah, and whether it’s your IT- and technical-knowledge-based workers in hospitals who facilitate all the technology โ we rely on an immigrant workforce for a lot of the technology sector. And then you’ve got research professionals. A lot of clinical researchers, medical researchers, are foreign-born. So it’s not just about the doctors. It’s also the critical staff that keep the hospitals operating. And I’m from Florida. For us, it’s the home health care workers. We have an aging population, and a large percentage of the home health care workers, particularly in Florida, happen to be Haitians on TPS [temporary protected status] or people with asylum work authorizations. And when we lose that, our aging population is left with no resources, because that’s not something AI or technology can fix. You can’t turn someone over in a bed with a robot yet, and we’re probably decades away from that.
Rovner: So what’s the last year been like for you and your clients?
Canero: I think it’s a lot of uncertainty. A lot of these policies are percolating, and we’re assuming that they’ll be resolved in litigation, but the damage is being done in real time. So we’re seeing hospitals turning away from hiring foreign workers, because of the H-1B penalty now. The suspension of J-1 processing created backlogs. These visa bans that affect 75 countries on certain visas and 39 countries on others. You’ve got thousands of health care workers that are stuck outside the U.S. So what’s happening, really, is that hospitals and medical providers are just shutting down, and they’re cutting back services, and that means that there are less available services and resources for the same population and the same demand. People are waiting longer for doctor’s appointments. People are finding that they’re not able to get to the specialist that they need to get to in time. And so for us as practitioners, I think, we’re trying to navigate as best we can, but we’re just seeing a lot of people, employers that traditionally would rely on our services, give up and foreign workers looking to go elsewhere.
Rovner: I noticed during the annual residency match in March that it worked out, I think, fairly well for most graduating medical students. But the big sort of sore thumb that stuck out were international medical graduates. That’s going to impact the pipeline going forward, isn’t it?
Canero: From what I understand, it takes like seven to 15 years to get to that level, and we just don’t have the student body to meet the demand of residency positions. From my understanding, there’s a gap between American graduates and the demand for residents that’s usually filled by foreign workers. And if we don’t have those foreign workers, those residency positions just don’t get filled. And that becomes more expensive for hospitals, and that transfers to our medical bills.
Rovner: And people assume that, Oh well this doesn’t impact me. But it really impacts all patients, doesn’t it? And I would think particularly those in rural areas, which are less desirable for U.S.-born and -trained medical professionals and tend to be overrepresented by immigrants.
Canero: Yeah, I think a lot of the J-1 doctors and H-1B doctors are what facilitate, are working at, our veterans hospitals and our rural medical facilities. And what’s ending up happening is the very same people that this administration touts to support their interests are being forced to travel farther for specialists, right? If there isn’t an endocrinologist in your area, you may have to drive 100 miles to go see that specialist, and you may forgo necessary medical care because of the inconvenience or the cost. And I think that’s hitting at our health.
Rovner: So you’re on the board of Immigrants’ List, which is working to change things politically. What’s one change that could really make a big difference in what we’re starting to see in terms of immigration and the health care workforce?
Canero: Well, asking Congress to actually do something. It’s been a problem for decades. So I don’t really know, but I think there’s a couple of things, whether it’s just policymakers supporting our fight against some of these illegal policy changes in courts, organizations supporting us with amicus briefs. For example, there’s a lot of lawsuits challenging these visa bans and these adjudicative holds and the H-1B fine. The more support that the plaintiffs in the litigation get, the more likely we are to resolve that through the court system. And then I hope that there’s enough pressure from hospitals and organizations that have real dollars that impact these elected officials to get them to start seeing, Hey, we need to pass reasonable immigration reform to address some of the loopholes that this administration is using to cause chaos in the system, right? They’re able to do this because we have a gap. We allow them to terminate TPS. We don’t have a structure to ensure that a community that’s been on TPS for 20 years gets grandfathered into some sort of more stable visa. We don’t have a system that precludes the administration from just putting a hold or a visa ban on nationalities. So it’s something that Congress is going to have to step up and do something about.
Rovner: What worries you most about sort of what’s going on with the immigration system and health care? What keeps you up at night? Obviously you, I know you work on more than just health care.
Canero: I think my concern is that the American people aren’t seeing what’s happening, or they’re sort of turning a blind eye to it, and by the time it starts to actually impact them and they start asking, Wait, wait, wait. Why is this happening? I don’t understand, it’s going to be too late. Because it’s not hitting their pocket, because it’s not their suffering at this point, they’re not standing up and saying, Hey, this needs to stop, at the level that we need, opposition, to make it stop. And by the time it does hit their pocket and it does affect them directly, I think, it’ll be a little too late. I think people will be scared off from coming here, people that we needed will be gone, and to reverse the system is going to take decades.
Rovner: Michelle Canero, thanks again.
Canero: No, you’re very welcome. Thank you for your time.
Carey: OK, we’re back. Now it’s time for our extra-credit segment, and that’s where we each recognize a story we read this week and we think that you should read it, too. Don’t worry if you miss it. We’ll post the links in our show notes. Joanne, why don’t you start us off this week?
Kenen: Well, this is by Teddy Rosenbluth in The New York Times. The headline is “” This is one of those stories where you know exactly how it’s going to end in the first paragraph, and yet it was so compellingly and beautifully written that you kept reading until the last word. It is, as the headline suggested, a young man who is an expert on AI and cognitive science named Ben Riley discovered that his father had been lying about a controllable, treatable form of leukemia. He had denied treatment, he’d refused treatment, he had ignored his oncologist because he was relying on AI. And as we all know, AI has its up moments and its down moments. And he was getting incorrect information, distrusted the diagnosis, refused treatment, getting sicker and sicker and sicker as the oncologist and the family got increasingly desperate. And the son, Ben Riley, had, like, skills. He knew how to find scientific evidence, and his father just would not believe it. And by the time his father finally consented to treatment, it was too late, and he did die. And his father was a neuroscientist, a retired neuroscientist, but he found a neuroscience rabbit hole.
Carey: That’s amazing. Anna, what’s your extra credit?
Edney: Mine, I’m highlighting a story that I wrote in Bloomberg called “.” And this is, I wanted to dive into this policy that the FDA had implemented. The commissioner has long talked about and felt that perimenopausal and menopausal women were not getting access to the treatments that maybe they really needed, because there had been sort of this two-decade-old study that had showed there were some safety issues regarding breast cancer and cardiovascular disease, but the issue being that those studies had looked at older forms of the medication and also at women who were much older than those who might benefit from taking it. And so they, the agency, asked the companies to remove those warning labels, at least the strongest ones. And what we’ve seen, why โ I wanted to dive into the numbers specifically. Bloomberg has some prescription data that was able to help me out here and just look at when this started rising. You could see that the prescriptions started going up around 2021. I feel like a lot of influencers, a lot of celebrities, were talking about this. And then in 2024 to 2025 when the FDA started talking about this, it really just goes, the prescription numbers just go straight up on the scale. And so there were about 32 million prescriptions written last year, which is a huge increase. And I just dove into some of this, some of the companies, what kind of drugs there are out there, and talked to some women who are benefiting but also, because of this pop, experiencing shortages, because the companies aren’t quite keeping up with the products.
Carey: Wow, that sounds like an outstanding deep dive. Thank you. Emmarie.
Huetteman: Yeah, my extra credit is from my colleague at ั๎น๓ๅ๚ดซรฝา๎l Health News who covers health technology. That’s Darius Tahir. The headline is “Your New Therapist: Chatty, Leaky, and Hardly Human.” The story looks at the proliferation of AI chatbot apps that offer mental health and emotional support, particularly the ones that market themselves as, quote-unquote, “therapy apps.” Darius counted 45 such apps in Apple’s App Store last month, and he uncovered in some cases that safety and privacy concerns existed, such as minimal age protections. Fifteen of the apps that he looked at said they could be downloaded by users who were only 4 years old. His story also explored the tension between the risks of sharing sensitive data and the interests of app developers and collecting that data for business purposes. It’s a good read. All right,
Carey: All right. Thanks so much. My extra credit is from Politico, and it’s written by Alice Miranda Olstein, and she’s a frequent guest here on What the Health? The headline is, quote, “,” close quote. The headline kind of says it all. Alice writes that Nebraska is racing to implement Medicaid work requirements by May 1, and that’s eight months ahead of the national deadline that was set by the One Big Beautiful Bill Act. Nebraska state officials plan to do this without hiring additional staff, even as other health departments in other states prepare to bring in dozens, if not hundreds, of new employees. Alice writes that advocates for people on Medicaid fear that this rush timeline and lack of new staff will cause many problems for Medicaid beneficiaries who are just trying to meet those new work requirements.
All right. That’s this week’s show. Thank you so much for listening. Thanks, as always, to our editor and panelist Emmarie Huetteman, to this week’s producer and engineer, Taylor Cook, and to my KFF colleague Richard Ho, who provided technical assistance. A reminder: What the Health? is now available on WAMU platforms, the NPR app, and wherever you get your podcasts, as well as, of course, kffhealthnews.org. Also, as always, you can email us with your comments or questions. We’re at whatthehealth@kff.org. Or you can find me on X, . Joanne, where can people find you these days?
Kenen: and , @joannekenen.
Carey: OK. Anna?
Edney: and and , @annaedney.
Carey: And Emmarie.
Huetteman: You can find me on .
Carey: We’ll be back in your feed next week. Until then, be healthy.